 Potassium chloride (KCl) is used in IR spectroscopy as well as in high power CO2 laser applications. It refers to salt crystals (like KBr and NaCl). KCl is a soft and stable to thermal influence monocrystal. Due to material hygroscopicity the polished parts require storage in desiccator or in sealed package with silica gel in a warm room.
Potassium chloride (KCl) is used in IR spectroscopy as well as in high power CO2 laser applications. It refers to salt crystals (like KBr and NaCl). KCl is a soft and stable to thermal influence monocrystal. Due to material hygroscopicity the polished parts require storage in desiccator or in sealed package with silica gel in a warm room.
Application:
- IR spectroscopy
- CO2 laser optics
Product types:
- Plane-parallel windows and wedges
- Lenses
Specifications
Tab.1. Typical specification of KCl optical components
| Specification |
Typical |
State-of-the-art |
| Sizes |
See table in the article Plane Windows and Wedged Windows |
Up to 100 mm |
| Diameter tolerance, mm |
+0/-0.25 |
RFQ |
| Thickness tolerance, mm |
±0.25 |
RFQ |
| Thickness matching, mm |
— |
RFQ |
| Surface quality, scr/dig |
60/40 |
RFQ |
| Surface flatness, λ @ 633nm per inch* |
2 |
RFQ |
| Parallelism (wedge tolerance) |
5 arc min |
RFQ |
| Coating |
none |
protecting |
| Packaging |
Typak® |
|
|
Transmission Spectrum
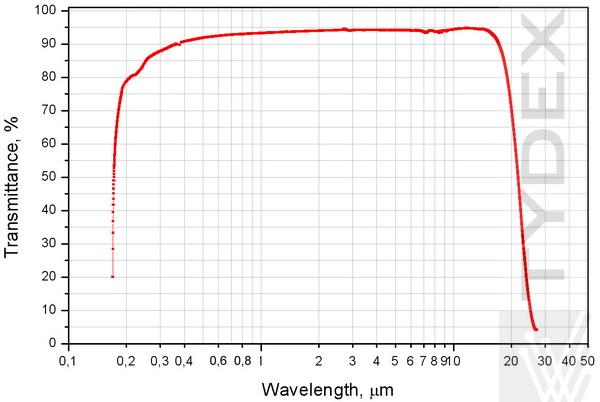
The measurements were carried out on Perkin Elmer Lambda-35 spectrophotometer and on BrukerVertex-70 Fourier-spectrometer.
Tab.2. Refractive index
| λ, µm |
n |
| 0.20 |
1.7187 |
| 0.51 |
1.4962 |
| 1.18 |
1.4783 |
| 5.00 |
1.4705 |
| 6.00 |
1.4684 |
| 7.00 |
1.466 |
| 8.00 |
1.4629 |
| 9.00 |
1.4600 |
| 10.00 |
1.4564 |
| 11.00 |
1.4524 |
| 12.00 |
1.4480 |
| 13.00 |
1.4431 |
| 14.00 |
1.4378 |
| 15.00 |
1.4320 |
| 16.00 |
1.4256 |
| 17.00 |
1.4188 |
| 18.00 |
1.4113 |
| 19.00 |
1.4033 |
| 20.00 |
1.3947 |
| 21.00 |
1.3854 |
| 22.00 |
1.3754 |
| 23.00 |
1.3646 |
| 24.00 |
1.3530 |
| 25.00 |
1.3406 |
| 26.00 |
1.3272 |
| 27.00 |
1.3128 |
| 28.00 |
1.2973 |
|
Tab.3. Optical properties
| Transmission range, microns |
0.22-21 |
| Colour |
Colourless |
| Reflection losses @ 11 µm (2 surfaces), % |
8.3 |
| Absorption coefficient, @ 3.8 µm, 10-4 cm-1 |
1.6 |
| Restrahlen peak, μm |
63.1 |
| dN/dT, 10-6/C |
-33.2 |
|
Tab.4. Physical and mechanical properties
| Class / Structure |
Cubic FCC, NaCl type, Fm3m, (100) cleavage |
| Density @300K, g/cm3 |
1.98 |
| Molecular Weight |
74.55 |
| Melting Point, °C |
776 |
| Thermal Conductivity @322K, W/(mxK) |
6.53 |
| Thermal Expansion @300K, 10-6/C |
36 |
| Hardness, Knoop with 200g indenter |
in (100) |
9.3 |
| in (110) |
7.2 |
| Specific Heat Capacity, J/(kg×K) |
690 |
| Debye temperature, K |
235 |
| Dielectric Constant for 106 Hz @300K |
4.64 |
| Bandgap, eV |
8.5 |
| Young Modulus (E), GPa |
29.67 |
| Shear Modulus (G), GPa |
6.24 |
| Bulk Modulus (K), GPa |
17.36 |
| Elastic Coefficient |
C11 = 40.2 C12 = 6.7 C44 = 6.29 |
| Apparent Elastic Limit, MPa |
2.3 (330psi) |
| Poisson Ratio |
0.216 |
|
Tab.5. Chemical stability / Solubility
in water
(at 0 оС) |
34.7 g/100cm3
hygroscopic |
| in acids |
soluble |
| in organic solvents: |
|
| spirits |
insignificantly soluble |
|
Please pay your attention that this article is for your information only. We do not supply KCl in ingots as well as semi-finished products. Our standard products are polished parts.
For further information on our KCl optical components please see the following: Windows for IR-spectroscopy, Packaging or fill in our request form.
 Potassium chloride (KCl) is used in IR spectroscopy as well as in high power CO2 laser applications. It refers to salt crystals (like KBr and NaCl). KCl is a soft and stable to thermal influence monocrystal. Due to material hygroscopicity the polished parts require storage in desiccator or in sealed package with silica gel in a warm room.
Potassium chloride (KCl) is used in IR spectroscopy as well as in high power CO2 laser applications. It refers to salt crystals (like KBr and NaCl). KCl is a soft and stable to thermal influence monocrystal. Due to material hygroscopicity the polished parts require storage in desiccator or in sealed package with silica gel in a warm room.






